 Open Access
Open Access
REVIEW
Targeting MDM2-p53 interaction for breast cancer therapy
1 Clinical Laboratory Sciences Department, College of Applied Medical Sciences, Taibah University, Madinah, 41477, Saudi Arabia
2 Institute of Biotechnology and Genetic Engineering, The University of Agriculture, Peshawar, Peshawar, 25130, Pakistan
* Corresponding Author: NAJEEB ULLAH KHAN. Email:
Oncology Research 2025, 33(4), 851-861. https://doi.org/10.32604/or.2025.058956
Received 25 September 2024; Accepted 19 December 2024; Issue published 19 March 2025
Abstract
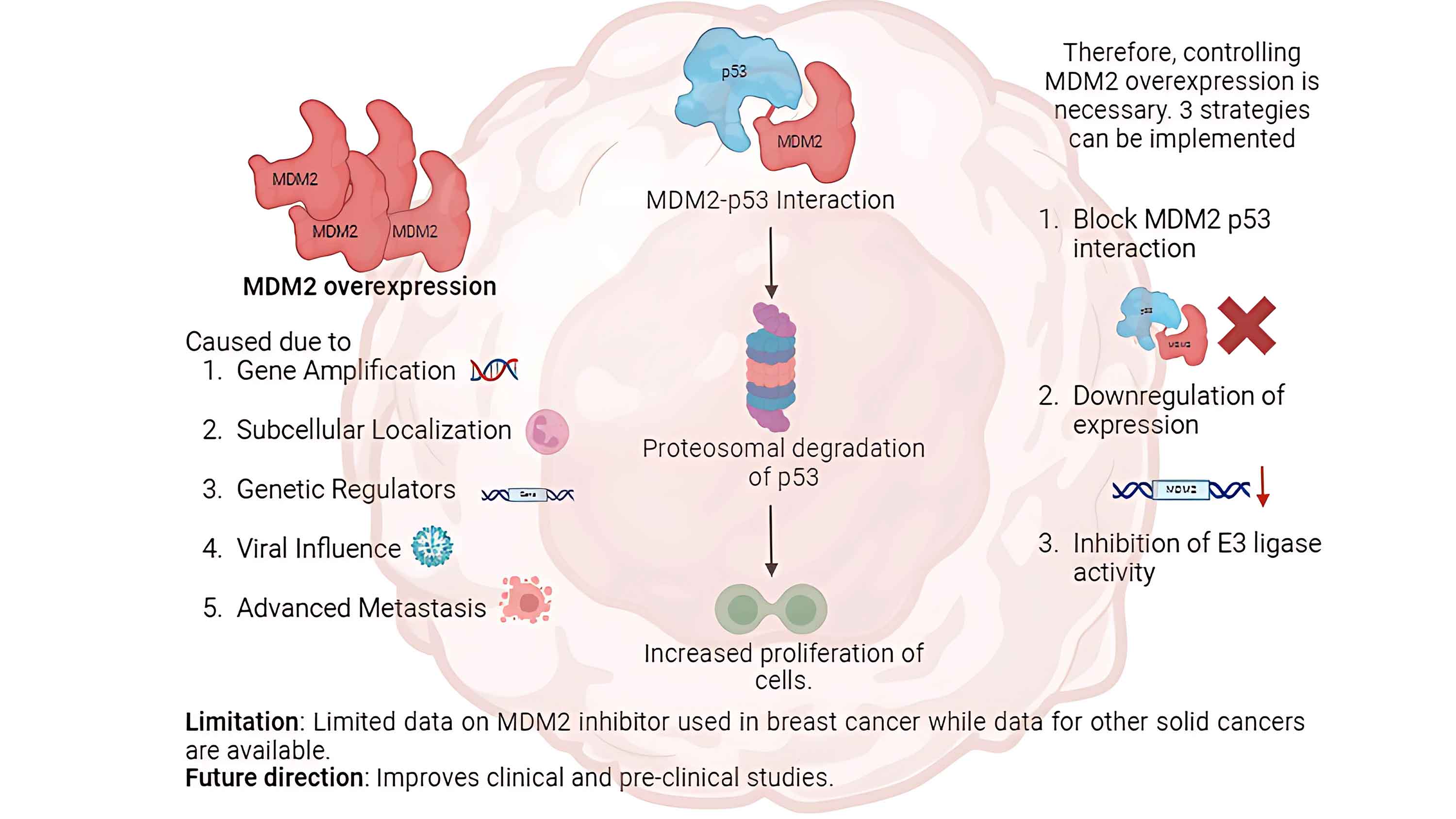
Breast cancer is a significant global concern, with limited effective treatment options. Therefore, therapies with high efficacy and low complications, unlike the existing chemotherapies, are urgently required. To address this issue, advances have been made in therapies targeting molecular pathways related to the murine double minute 2 proto-oncogene (MDM2)-tumor proteinp53 (TP53) interaction. This review aims to investigate the efficacy of MDM2 inhibition in restoring TP53 activity in breast cancer cells, as evidenced by clinical studies, reviews, and trials. TP53 is a tumor suppressor and MDM2 facilitates proteasomal degradation of TP53. MDM2 and TP53 activity is tightly regulated. However, cancerous breast cells overexpress MDM2 through five hypothesized mechanisms. Consequently, TP53 levels decrease with increased tumor cell proliferation. Three strategies have been identified for controlling MDM2 upregulation in cells with wild-type or mutated TP53. MDM2 inhibitors (MDM2i) are administered in combination with existing chemotherapies to reduce their effects on healthy cells. Few clinical and preclinical studies have been conducted using MDM2i, which necessitates high-quality clinical trials to support their therapeutic potential in breast cancer therapy.Graphic Abstract
Keywords
Cite This Article
 Copyright © 2025 The Author(s). Published by Tech Science Press.
Copyright © 2025 The Author(s). Published by Tech Science Press.This work is licensed under a Creative Commons Attribution 4.0 International License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.


 Submit a Paper
Submit a Paper Propose a Special lssue
Propose a Special lssue View Full Text
View Full Text Download PDF
Download PDF Downloads
Downloads
 Citation Tools
Citation Tools